Early development of the heart:
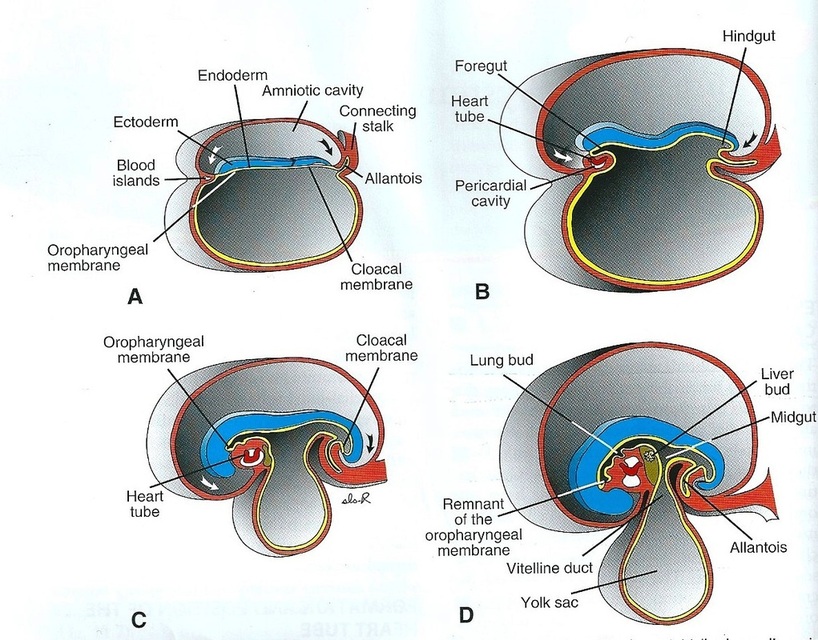
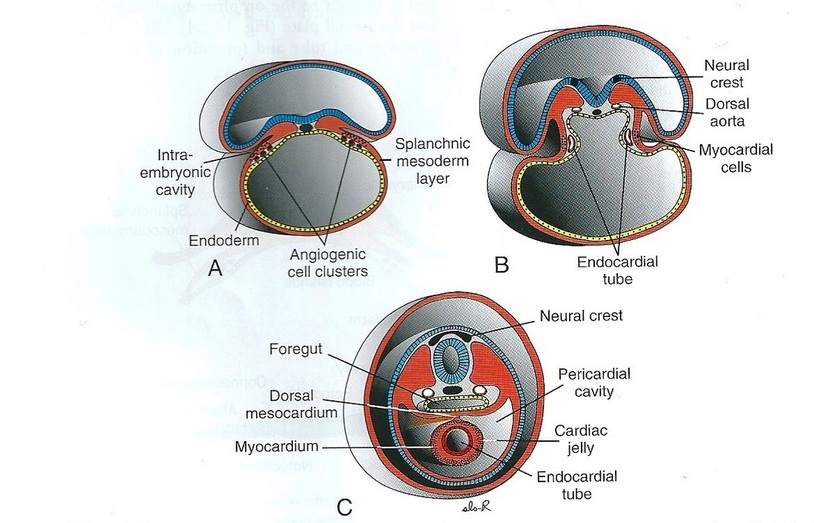
First, the central portion of the cardiogenic area is anterior to the oropharyngeal membrane and the neural plate. Although it is close to the neural tube and formation of the brain vesicles, the central nervous system extends over the central cardiogenic area and the future pericardial cavity (Fig.2). The oropharyngeal membrane pulls forward, while the heart and pericardial cavity move to the cervical region and then to the thorax due to brain growth and cephalic folding. Because the embryo folds cephalocaudally and laterally, the caudal regions of the paired cardiac primordia are merged except at their caudalmost ends. The future outflow tract and ventricular regions are formed by the crescent part of the horseshoe-shaped area. Consequently, the heart has an inner endothelial lining and an outer myocardial layer. It receives venous drainage at its caudal pole and begins to pump blood out of the first aortic arch into the dorsal aorta at its cranial pole. Although the heart tube bulges into the pericardial cavity, it remains attached to the dorsal side of the pericardial cavity by a fold of mesodermal tissue, the dorsal mesocardium, which disappears with further development and creation of the transverse pericardial sinus that connects both sides of the pericardial cavity. No ventral mesocardium is formed. In the cavity, the blood vessels at the cranial and caudal poles suspend the heart. During heart development, the heart tube consists of three layers. The first layer is the endocardium, which forms the internal endothelial lining of the heart. The second is the myocardium, which forms the muscular wall. The third is the epicardium or visceral pericardium, which covers the outside of the tube and is responsible for the formation of the coronary arteries, including their endothelial linings and smooth muscle (Fig.3).
Figure.2: Effects of rapid growth of the brain on heart position (Sadler, 2009).
Figure.3: Formation of the heart tube (Sadler, 2009).
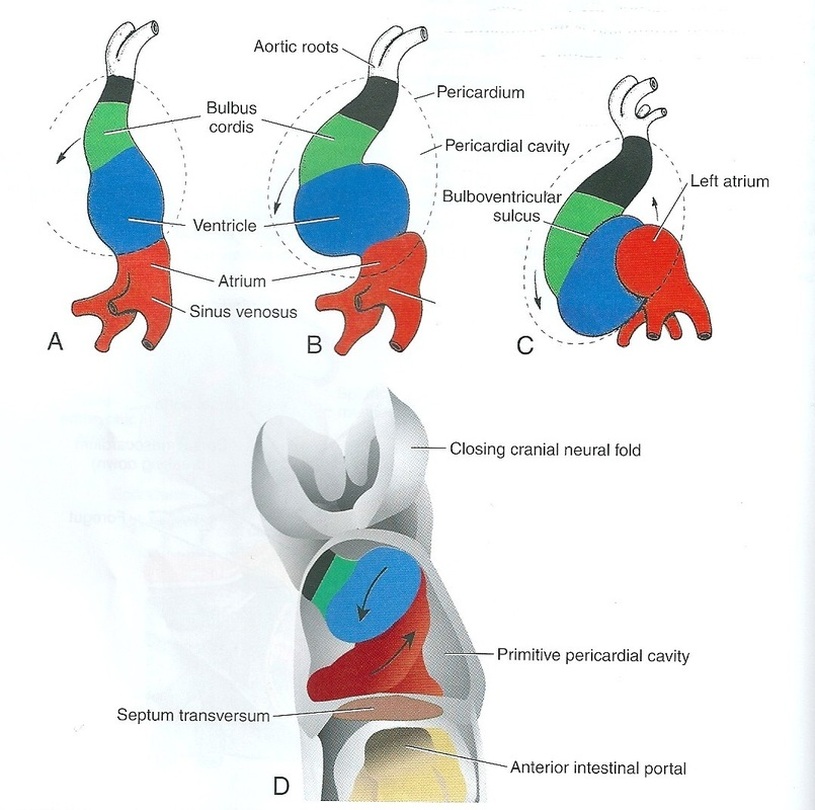
Formation of the cardiac loop (Fig.4):
On day 23, the folding of the heart tube starts to change the tube from anterior/posterior polarity to left/right polarity. This is an unknown mechanism; however, factor Pitx2 is responsible for differentiating the heart tube during embryonic development. It depends on heart-specific proteins, which are activated by Nkx2.5 such as Hand1, Hand2, and Xin. Although the heart folding occurs after the heart chambers, it cannot be distinguished after folding.
The common atrium and the atrioventricular canal are formed by constriction at the atrioventricular junction. In addition, the proximal third of the bulbus cordis forms the trabeculated part of the right ventricle. The midportion is the conus cordis, which forms the outflow tracts of the ventricles. The last part is the distal part, which is known as thetruncus arteriosus and forms the roots of the aorta and pulmonary artery. Externally, the bulboventricular sulcus indicates the junction between the ventricle and the bulbus cordis and is known as the primary interventricular foramen.
At the end of the loop formation, just proximal and distal to the interventricular foramen, primitive trabeculae begin to form: the bulbus cordis remains smooth-walled. Then, the primitive left ventricle forms from the trabeculated primitive ventricle and the trabeculated proximal third of the bulbus cordis may form the primitive right ventricle.
The common atrium and the atrioventricular canal are formed by constriction at the atrioventricular junction. In addition, the proximal third of the bulbus cordis forms the trabeculated part of the right ventricle. The midportion is the conus cordis, which forms the outflow tracts of the ventricles. The last part is the distal part, which is known as thetruncus arteriosus and forms the roots of the aorta and pulmonary artery. Externally, the bulboventricular sulcus indicates the junction between the ventricle and the bulbus cordis and is known as the primary interventricular foramen.
At the end of the loop formation, just proximal and distal to the interventricular foramen, primitive trabeculae begin to form: the bulbus cordis remains smooth-walled. Then, the primitive left ventricle forms from the trabeculated primitive ventricle and the trabeculated proximal third of the bulbus cordis may form the primitive right ventricle.
Figure.4: Formation of the cardiac loop (Sadler, 2009).
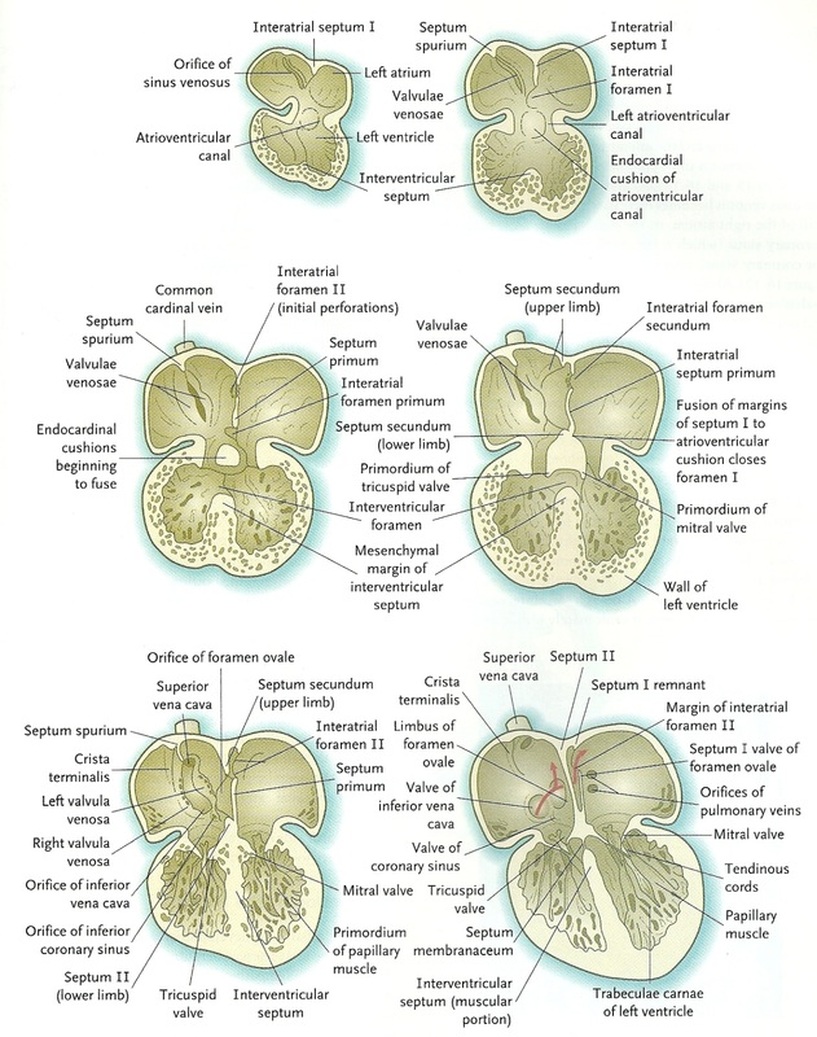
Later partitioning of the heart (Fig.5):
If the heart had separate chambers in the early weeks of fetus life, the pulmonary circulation would be overstressed; in addition, the left side might not pump enough blood to the other systems.
Separation of the atria from the ventricles:
The endocardial cushions are formed on the dorsal and ventral walls of the atrioventricular canal, and the two cushions meet to separate the atrioventricular canal into right and left channels In early development, primitive valves can assist forward propulsion of the blood through the heart, while in the later stages of heart development there are thin leaflets of anatomical valves in the atrioventricular canal. The valve that protects the right atrioventricular canal develops three leaflets known as the tricuspid valve. The valve that develops only two leaflets in the left canal is known as the mitral or bicuspid valve.
Partition of the atria:
This starts in the fifth week following fertilisation when a crescentic interatrial septum primum grows down from the cephalic wall of the common atrium extending toward the atrioventricular canal to merge with the endocardial cushions. There is a space, known as the interatrial foramen primum, between the septum primum and the endocardial cushions to permit blood to pass directly from the right to the left atrium. When obliterating the foramen primum during fusing between the edge of the septum primum and the endocardial cushions, cephalic perforations appear due to genetically programmed cell death and give rise to the interatrial foramen secundum, a new foramen that preserves the connection between the right and left atria. After that, the septum secundum grows out from the dorsal to the ventral part of the atrium just to the right of the septum primum: it forms the foramen ovale which allows most of the blood that enters the right atrium via the inferior vena cava to pass into the left atrium. This acts as a one-way valve that permits blood to flow from the right to the left atrium, but not in the opposite direction.
Partitioning of the ventricles:
Amuscular interventricular septum grows from the apex of the common ventricle toward the atrioventricular endocardial cushions during formation of the interatrial septa. Initially, on interventricular foramen is present; however, this is ultimately obliterated. This occurs with further growth of the muscular interventricular septum, a contribution by truncoconal ridge tissue which divides the outflow tract of the heart, and a membranous component derived from endocardial cushion connective tissue.
Separation of the atria from the ventricles:
The endocardial cushions are formed on the dorsal and ventral walls of the atrioventricular canal, and the two cushions meet to separate the atrioventricular canal into right and left channels In early development, primitive valves can assist forward propulsion of the blood through the heart, while in the later stages of heart development there are thin leaflets of anatomical valves in the atrioventricular canal. The valve that protects the right atrioventricular canal develops three leaflets known as the tricuspid valve. The valve that develops only two leaflets in the left canal is known as the mitral or bicuspid valve.
Partition of the atria:
This starts in the fifth week following fertilisation when a crescentic interatrial septum primum grows down from the cephalic wall of the common atrium extending toward the atrioventricular canal to merge with the endocardial cushions. There is a space, known as the interatrial foramen primum, between the septum primum and the endocardial cushions to permit blood to pass directly from the right to the left atrium. When obliterating the foramen primum during fusing between the edge of the septum primum and the endocardial cushions, cephalic perforations appear due to genetically programmed cell death and give rise to the interatrial foramen secundum, a new foramen that preserves the connection between the right and left atria. After that, the septum secundum grows out from the dorsal to the ventral part of the atrium just to the right of the septum primum: it forms the foramen ovale which allows most of the blood that enters the right atrium via the inferior vena cava to pass into the left atrium. This acts as a one-way valve that permits blood to flow from the right to the left atrium, but not in the opposite direction.
Partitioning of the ventricles:
Amuscular interventricular septum grows from the apex of the common ventricle toward the atrioventricular endocardial cushions during formation of the interatrial septa. Initially, on interventricular foramen is present; however, this is ultimately obliterated. This occurs with further growth of the muscular interventricular septum, a contribution by truncoconal ridge tissue which divides the outflow tract of the heart, and a membranous component derived from endocardial cushion connective tissue.
Figure.5: Partitioning of the heart (Sadler, 2009).
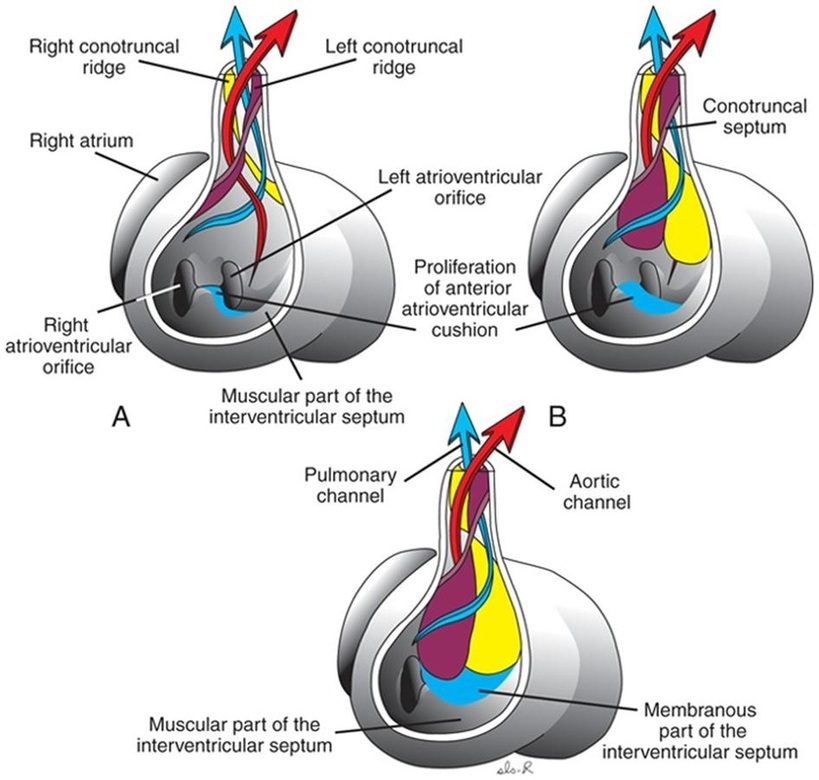
Partitioning of the outflow tract of the heart (Fig.6):
The outflow tract is a single tube (bulbus cordis) in the early stages; however, with time the bulbus elongates and divides into a proximal conus arteriosus and a distal truncus arteriosus. Through the appearance of two spiral truncoconal ridges, the outflow tract is partitioned into aortic and pulmonary channels. These ridges, which are derived from neural crest mesenchyme, bulge into the lumen and finally meet, separating it into two channels. This partitioning starts near the ventral aortic root between the fourth and sixth arches and extends to the ventricles, spiraling as it goes. Two new sets of semilunar valves, which each have three leaflets, form at the base of the conus. These valves prevent ejected blood from flowing back into the ventricles.
Figure.6: Partitioning of the outflow tract of the heart (Sadler, 2009).
Copyright © 2011 by Reem Bu Saeed.
Last updated: 30-07-2011.
Last updated: 30-07-2011.